On the evening of March 29, 2026, Insilico Medicine (“Insilico”, HKEX: 3696), a clinical-stage biotechnology company powered by generative artificial intelligence (AI) and automation, announced its annual results for 2025. During the Reporting Period, Insilico Medicine further expanded the value delivery framework of its "AI + Drug Discovery" dual-engine model through three key areas: continuously enhancing its AI platform capabilities, advancing its proprietary pipeline at scale, and achieving rapid growth in business partnerships and milestone payments.
During the Reporting Period, the Company generated total revenue of US$56.24 million, demonstrating diversified contributions across business segments. On December 30, 2025, Insilico Medicine successfully listed on the Main Board of the Stock Exchange of Hong Kong, receiving recognition from global investors including Eli Lilly and Tencent. The IPO became Hong Kong's largest biotech fundraising for the year, bringing the Company's cash and bank balances to US$393.3 million as of December 31, 2025, providing strong capital support for operations and advancing innovation projects.
Pharma.AI Upgrades Drive Commercial Growth:
Serving 13 of Top 20 Global Pharmaceutical Companies, Software Revenue +23.8%
During the Reporting Period and as of the Latest Practicable Date*, Insilico Medicine continued to upgrade and expand Pharma.AI, its proprietary end-to-end generative AI platform, completing comprehensive upgrades across Biology42, Chemistry42 and Science42.
Leveraging high-quality data accumulation and technological advantages, the Company completed foundational technical construction including target identification frameworks, chemistry foundation models and MMAI Gym training framework, further supporting the Company's strategic goal of advancing toward "pharmaceutical superintelligence."
In terms of commercialization, the Company's software platform serves 13 of the top 20 global pharmaceutical companies**; software revenue in 2025 increased 23.8% year-over-year, with subscription customer base growing 18.3% year-over-year.
Leveraging high-quality data accumulation and technological advantages, the Company completed foundational technical construction including target identification frameworks, chemistry foundation models and MMAI Gym training framework, further supporting the Company's strategic goal of advancing toward "pharmaceutical superintelligence."
In terms of commercialization, the Company's software platform serves 13 of the top 20 global pharmaceutical companies**; software revenue in 2025 increased 23.8% year-over-year, with subscription customer base growing 18.3% year-over-year.
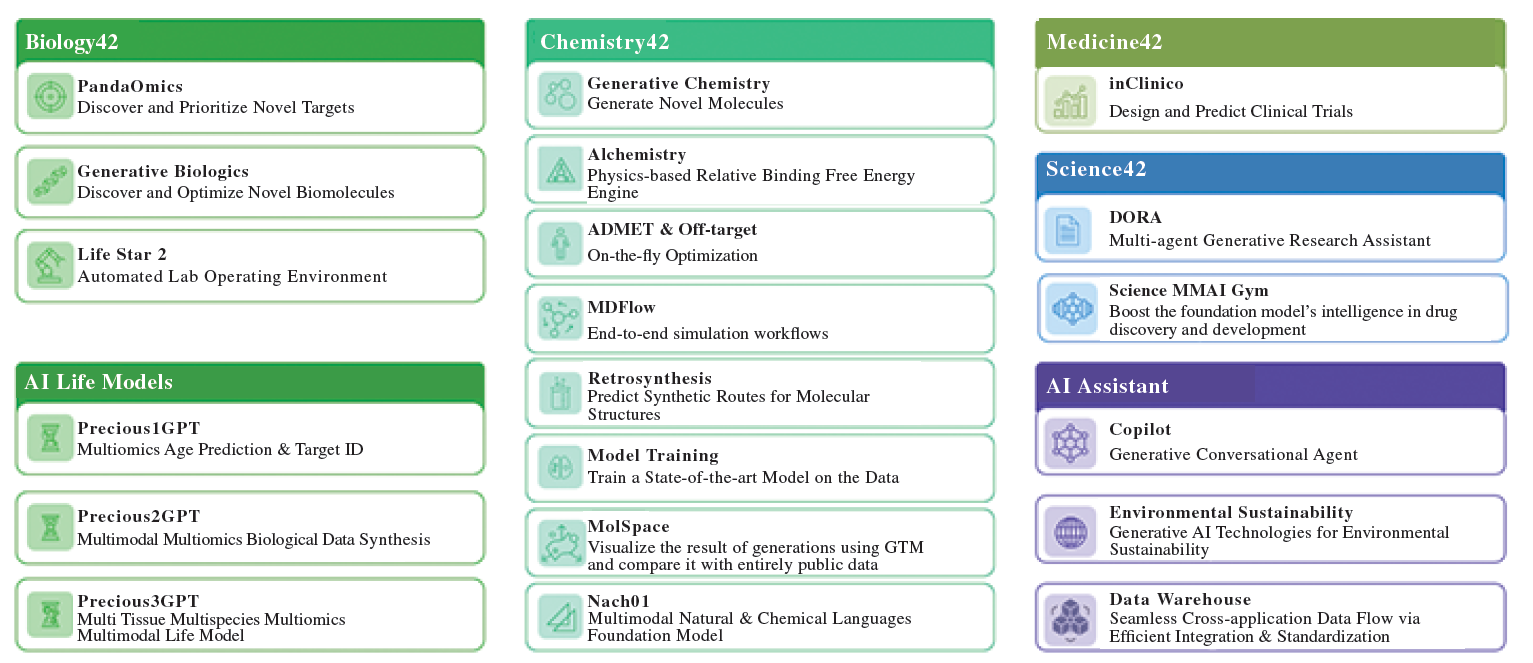
Biology42
The target discovery engine PandaOmics completed core functional upgrades, strengthening multi-dimensional screening criteria including confidence, commercial tractability, druggability, and mechanism clarity. Also, working alongside TargetPro, a specialized target identification framework, the Company launched TargetBench 1.0—the industry's first drug target discovery benchmark. Together, these platforms establish a complete loop from hypothesis generation through validation to assessment.
For biologics, the Generative Biologics platform improved PDB integration and introduced a diffusion-based antibody design engine, significantly improving R&D efficiency for both peptide and antibody molecules.
The target discovery engine PandaOmics completed core functional upgrades, strengthening multi-dimensional screening criteria including confidence, commercial tractability, druggability, and mechanism clarity. Also, working alongside TargetPro, a specialized target identification framework, the Company launched TargetBench 1.0—the industry's first drug target discovery benchmark. Together, these platforms establish a complete loop from hypothesis generation through validation to assessment.
For biologics, the Generative Biologics platform improved PDB integration and introduced a diffusion-based antibody design engine, significantly improving R&D efficiency for both peptide and antibody molecules.
Chemistry42
The platform enhanced critical capabilities in ADMET assessment and off-target risk prediction, while specialized engines like Alchemistry continue to power innovative pipeline development. A major new addition is Nach01, a multimodal chemistry foundation model trained on billions of molecular and textual data points. Nach01 excels at molecular design and scientific reasoning, seamlessly processing both natural language and chemical structure information. The model is now available on AWS Marketplace and Microsoft Discovery platform for broad access.
Science42
Supported by Microsoft Azure, Insilico Medicine open-sourced and upgraded DORA, the multi-agent assistant designed for scientific research and content creation. DORA accelerates the research process through high-quality source retrieval, smart prompt combinations, and streamlined content generation workflows, making academic paper and scientific document writing far more efficient.
Science MMAI Gym
The Company also launched Science MMAI Gym, a foundation model training framework that fine-tunes models with causal reasoning capabilities into high-performance engines for real-world drug discovery and development. This framework is already delivering results. In March 2026, Insilico Medicine and Liquid AI completed the first MMAI Gym-based collaboration, achieving state-of-the-art (SOTA) performance using a lightweight model on private infrastructure, further unleashing AI application potential.
The platform enhanced critical capabilities in ADMET assessment and off-target risk prediction, while specialized engines like Alchemistry continue to power innovative pipeline development. A major new addition is Nach01, a multimodal chemistry foundation model trained on billions of molecular and textual data points. Nach01 excels at molecular design and scientific reasoning, seamlessly processing both natural language and chemical structure information. The model is now available on AWS Marketplace and Microsoft Discovery platform for broad access.
Science42
Supported by Microsoft Azure, Insilico Medicine open-sourced and upgraded DORA, the multi-agent assistant designed for scientific research and content creation. DORA accelerates the research process through high-quality source retrieval, smart prompt combinations, and streamlined content generation workflows, making academic paper and scientific document writing far more efficient.
Science MMAI Gym
The Company also launched Science MMAI Gym, a foundation model training framework that fine-tunes models with causal reasoning capabilities into high-performance engines for real-world drug discovery and development. This framework is already delivering results. In March 2026, Insilico Medicine and Liquid AI completed the first MMAI Gym-based collaboration, achieving state-of-the-art (SOTA) performance using a lightweight model on private infrastructure, further unleashing AI application potential.
Pipeline Expansion Accelerated by AI Platform:
6 Preclinical Candidates Nominated, 8 Programs Advanced in Clinical Development
Leveraging its AI platform, Insilico Medicine significantly expanded its pipeline, building a diverse product portfolio from preclinical through Phase II stages, spanning multiple therapeutic areas including fibrosis, oncology, immunology, metabolic diseases, and pain.
During the Reporting Period and as of the Latest Practicable Date, the Company nominated 6 new preclinical candidates and advanced 8 programs in clinical development. These achievements validate both the scalability of the AI platform and enhance opportunities for out-licensing and partnerships.
To date, Insilico Medicine has nominated 28 preclinical candidates in total, with 10 programs currently in clinical trials.
Preclinical Candidate Discovery
During the Reporting Period and as of the Latest Practicable Date, the Company nominated 6 preclinical candidates, including 4 publicly disclosed wholly-owned programs and 2 undisclosed collaboration programs.
Highlights of proprietary programs:
Clinical Stage Program Advancement
During the Reporting Period and as of the Latest Practicable Date, the Company advanced 8 programs in clinical development, including 4 proprietary programs and 3 collaboration programs; progress include 2 Phase II programs, 4 Phase I programs, and 2 IND milestones:
The complete study results were published in Nature Medicine and selected for multiple oral presentations at the 2025 American Thoracic Society (ATS) International Conference and Pulmonary Fibrosis Foundation Summit (PFF Summit 2025).
During the Reporting Period and as of the Latest Practicable Date, the Company nominated 6 new preclinical candidates and advanced 8 programs in clinical development. These achievements validate both the scalability of the AI platform and enhance opportunities for out-licensing and partnerships.
To date, Insilico Medicine has nominated 28 preclinical candidates in total, with 10 programs currently in clinical trials.
Preclinical Candidate Discovery
During the Reporting Period and as of the Latest Practicable Date, the Company nominated 6 preclinical candidates, including 4 publicly disclosed wholly-owned programs and 2 undisclosed collaboration programs.
Highlights of proprietary programs:
- ISM3830, An oral, highly selective small molecule CBLB inhibitor
- ISM0676, an innovative oral small molecule GIPR antagonist
- ISM5059, an innovative peripherally restricted small molecule NLRP3 inhibitor
- ISM6166, an innovative oral broad-spectrum pan-KRAS inhibitor
Clinical Stage Program Advancement
During the Reporting Period and as of the Latest Practicable Date, the Company advanced 8 programs in clinical development, including 4 proprietary programs and 3 collaboration programs; progress include 2 Phase II programs, 4 Phase I programs, and 2 IND milestones:
- ISM001-055 (Rentosertib), a small molecule TNIK inhibitor for treating idiopathic pulmonary fibrosis (IPF). The Phase IIa GENESIS-IPF study demonstrated strong safety and tolerability, with a dose-dependent improvement in lung function. This represents the first clinical proof-of-concept for an AI-discovered drug.
The complete study results were published in Nature Medicine and selected for multiple oral presentations at the 2025 American Thoracic Society (ATS) International Conference and Pulmonary Fibrosis Foundation Summit (PFF Summit 2025).
- ISM5411 (Garutadustat), a gut-restricted small molecule PHD1/2 inhibitor for inflammatory bowel disease (IBD) treatment. A Phase IIa clinical study named BETHESDA was launched, where first patient dosing was completed.
- ISM6331, a non-covalent small molecule pan-TEAD inhibitor for solid tumor treatment. Preclinical data showed broad-spectrum anti-tumor activity at low doses with favorable safety. Global multicenter Phase I trial initiated with first patient dosed.
- ISM3412, a small molecule MAT2A inhibitor for MTAP-deleted tumor treatment; preclinical studies demonstrated ideal efficacy, selectivity, druggability and combination potential. Initiated global multicenter Phase I trial and completed first patient dosing.
- ISM001-055 (Rentosertib)-Inhalation, an inhalable formulation of the TNIK inhibitor for treating idiopathic pulmonary fibrosis. Features potential advantages of high technical barriers, low efficacious dose, large safety window, and minimal side effects. IND application has been submitted for clinical trial.
Dual-driven Business Development Momentum:
10+ New Collaborations Totaling US$1.3 Billion, Multiple Programs Achieved Milestones
The Company's "AI + Drug Discovery" dual-engine model continued driving strong business development. During the Reporting Period and as of the Latest Practicable Date, Insilico Medicine established collaborations with over 10 multinational pharmaceutical companies and leading domestic biopharmaceutical firms across drug discovery, out-licensing, and other areas. Newly signed agreements totaled US$1.3 billion, bringing cumulative collaboration value to US$4.6 billion.
Beyond new deals, multiple existing programs reached key milestones, triggering corresponding milestone payments. This track record demonstrates the Company's ability to consistently deliver value through its platform and pipeline. The "licensing + milestone payments" model also strengthens cash flow visibility and supports larger-scale global partnerships and pipeline advancement.
Major Disclosed Collaborations Include:
ISM4808: Achieved Greater China region pipeline licensing collaboration with TaiGen Biotechnology.
Granted TaiGen and its wholly-owned Beijing subsidiary exclusive rights to develop, commercialize, and sublicense ISM4808 across Greater China, with a total deal size of two-digit million dollars.
ISM8969: Reached exclusive licensing and collaboration agreement with Hygtia Therapeutics.
Jointly advance global development of ISM8969, with both parties holding 50% global rights. Insilico Medicine is eligible to receive over USD$10 million in upfront and milestone payments.
Moreover, the Company established drug discovery collaborations with Eli Lilly, Hisun Pharma, Servier, Qilu Pharmaceutical, China Medical System, among others; reached target discovery collaborations with Memorial Sloan Kettering Cancer Center (MSK) and ASKA Pharma, achieved milestone delivery in drug discovery collaborations with Therasid Bio, Hisun Pharma and others, and received milestone payments in pipeline out-licensing or co-development collaborations with Menarini, TaiGen and others.
Beyond new deals, multiple existing programs reached key milestones, triggering corresponding milestone payments. This track record demonstrates the Company's ability to consistently deliver value through its platform and pipeline. The "licensing + milestone payments" model also strengthens cash flow visibility and supports larger-scale global partnerships and pipeline advancement.
Major Disclosed Collaborations Include:
ISM4808: Achieved Greater China region pipeline licensing collaboration with TaiGen Biotechnology.
Granted TaiGen and its wholly-owned Beijing subsidiary exclusive rights to develop, commercialize, and sublicense ISM4808 across Greater China, with a total deal size of two-digit million dollars.
ISM8969: Reached exclusive licensing and collaboration agreement with Hygtia Therapeutics.
Jointly advance global development of ISM8969, with both parties holding 50% global rights. Insilico Medicine is eligible to receive over USD$10 million in upfront and milestone payments.
Moreover, the Company established drug discovery collaborations with Eli Lilly, Hisun Pharma, Servier, Qilu Pharmaceutical, China Medical System, among others; reached target discovery collaborations with Memorial Sloan Kettering Cancer Center (MSK) and ASKA Pharma, achieved milestone delivery in drug discovery collaborations with Therasid Bio, Hisun Pharma and others, and received milestone payments in pipeline out-licensing or co-development collaborations with Menarini, TaiGen and others.
Towards Pharmaceutical Superintelligence, Empowering Healthspan Extension
Based on its proprietary Pharma.AI platform, Insilico Medicine looks forward to driving continuous integration and advancements across high-quality data, foundation models, and agent systems, further enhancing efficiency and success rates throughout the drug R&D process.
For wholly-owned pipelines, the Company will accelerate key clinical milestones for core programs while expanding its product portfolio to cover more indications. For business development, Insilico Medicine will broaden its partner network and deepen project engagement through diversified models, including software services, drug discovery services, and pipeline out-licensing, strengthening both revenue visibility and cash flow stability.
Through the continued advancements of AI capabilities, clinical validation, and collaboration milestones, Insilico Medicine is transforming its technical advantage of "faster preclinical development" into a commercial strength of "more predictable value delivery." In doing so, the Company is establishing a replicable growth model for the AI drug discovery industry and driving scalable, predictable commercialization.
Insilico Medicine will report its financial results for the year in live conference calls on March 30, 2026, where the management team will discuss 2025 operational and financial performance, R&D pipeline and key clinical progress, collaboration and commercialization progress, and 2026 business outlook, followed by a Q&A session with investors and analysts.
For wholly-owned pipelines, the Company will accelerate key clinical milestones for core programs while expanding its product portfolio to cover more indications. For business development, Insilico Medicine will broaden its partner network and deepen project engagement through diversified models, including software services, drug discovery services, and pipeline out-licensing, strengthening both revenue visibility and cash flow stability.
Through the continued advancements of AI capabilities, clinical validation, and collaboration milestones, Insilico Medicine is transforming its technical advantage of "faster preclinical development" into a commercial strength of "more predictable value delivery." In doing so, the Company is establishing a replicable growth model for the AI drug discovery industry and driving scalable, predictable commercialization.
Insilico Medicine will report its financial results for the year in live conference calls on March 30, 2026, where the management team will discuss 2025 operational and financial performance, R&D pipeline and key clinical progress, collaboration and commercialization progress, and 2026 business outlook, followed by a Q&A session with investors and analysts.
Conference Call Details:
For English Session:
Time: 9:00 AM Beijing Time, Monday, March 30, 2026
(9:00 PM U.S. Eastern Time, Sunday, March 29, 2026)
Register and Webcast Link:
https://insilico.zoom.us/w/81195639919?pwd=i153PUfkONs5INk8mgvLmmttveLqc0.1
Participants are required to pre-register for the English conference call using the link above. After registration is approved, participants will receive the webinar access link and dial-in information via email.
For Mandarin Session:
Time: 10:30 AM Beijing Time, Monday, March 30, 2026
(10:30 PM U.S. Eastern Time, Sunday, March 29, 2026)
Register and Webcast Link:
https://research.citics.com/tel/info/1_2047925
Participants are required to pre-register for the Mandarin conference call using the link above.
Alternatively, participants may dial in to the conference call using below dial-in information:
Mainland China 4008108228
Hong Kong, China 85230051313
International 861058084166
Singapore 6568185374
United States 16462543594
United Kingdom 441213680466
Password 977326
The replay of English session will be available shortly after the call and can be accessed by visiting the Company’s website at https://insilico.com/.
For English Session:
Time: 9:00 AM Beijing Time, Monday, March 30, 2026
(9:00 PM U.S. Eastern Time, Sunday, March 29, 2026)
Register and Webcast Link:
https://insilico.zoom.us/w/81195639919?pwd=i153PUfkONs5INk8mgvLmmttveLqc0.1
Participants are required to pre-register for the English conference call using the link above. After registration is approved, participants will receive the webinar access link and dial-in information via email.
For Mandarin Session:
Time: 10:30 AM Beijing Time, Monday, March 30, 2026
(10:30 PM U.S. Eastern Time, Sunday, March 29, 2026)
Register and Webcast Link:
https://research.citics.com/tel/info/1_2047925
Participants are required to pre-register for the Mandarin conference call using the link above.
Alternatively, participants may dial in to the conference call using below dial-in information:
Mainland China 4008108228
Hong Kong, China 85230051313
International 861058084166
Singapore 6568185374
United States 16462543594
United Kingdom 441213680466
Password 977326
The replay of English session will be available shortly after the call and can be accessed by visiting the Company’s website at https://insilico.com/.
*As defined in Insilico Medicine's annual results announcement for the year ended December 31, 2025, disclosed on March 29, 2026.
**In terms of reported sales in 2024.
**In terms of reported sales in 2024.
