QPCTL
A Small Molecule Inhibitor of QPCTL as a Potential Treatment of Tumors with High Engagement of CD47-Signal Regulatory Protein α ("SIRPα") Axis
-
Current Stage:
Phase I -
Co-development with Fosun Pharma

Indication
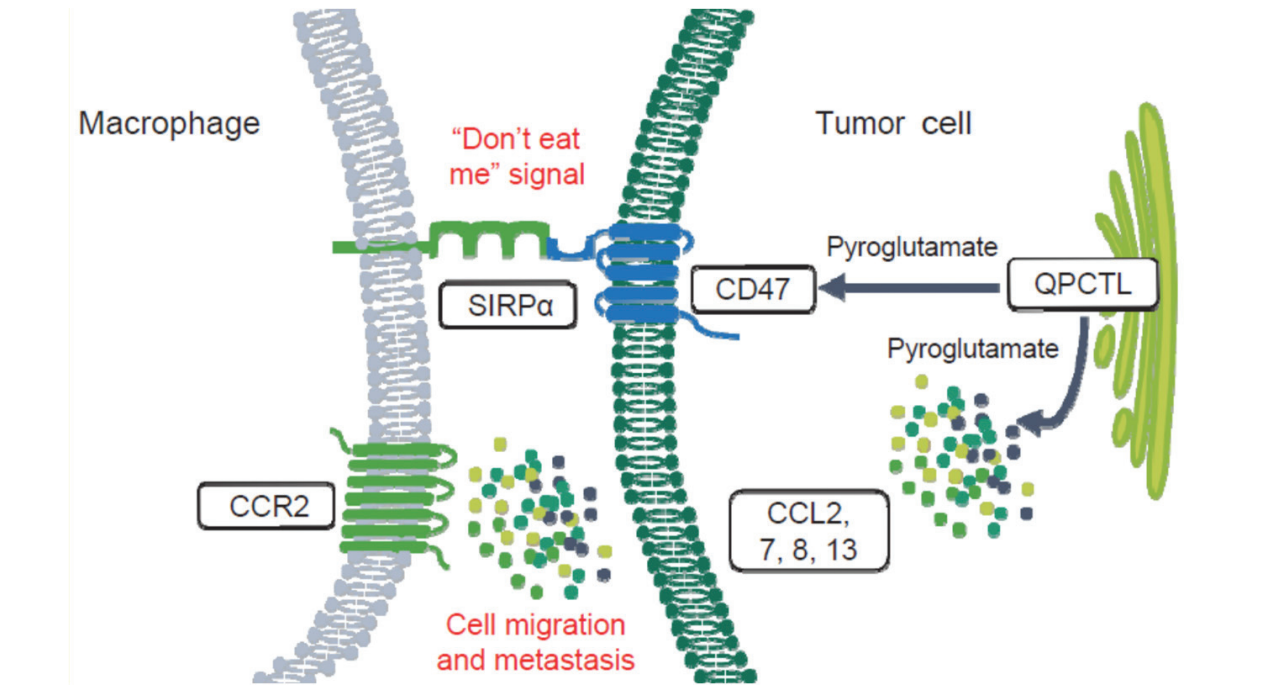
The detection and clearance of cancer cells via phagocytosis induced by innate immune checkpoints plays a significant role in tumor-mediated immune escape. The CD47-SIRPα axis, whose signaling is sustained by QPCTL, is the most well-described innate immune checkpoint which allows the CD47-expressing cancer cells to evade innate immune cell-mediated phagocytosis. The agents that block the CD47–SIRPα interaction are currently being evaluated in multiple ongoing trials, most in combination with pro-phagocytosis agents, e.g. therapeutic antibodies, chemotherapy in patients with leukemia, lymphoma and advanced solid tumors (head and neck squamous cell carcinoma, gastric cancer, breast cancer, colorectal cancer, etc.). The QPCTL inhibitor could be developed for the patients with these indications, especially when they are resistant to the standard-of-care therapies, e.g. azacytidine, rituximab, paclitaxel, trastuzumab, cetuximab, etc.
About ISM8207
ISM8027 is a novel, potent, orally bioavailable QPCTL inhibitor. In January 2022, Insilico Medicine announced a strategic collaboration with Fosun Pharma, with QPCTL inhibitor as one of the most advanced programs in the framework of the collaboration.
In preclinical studies, ISM8207 demonstrated a significant synergistic anti-tumor effect, as a combination agent, against multiple solid tumor and hematologic malignancy models. It also exhibits a favorable in vitro ADME and in vivo PK profile.
In preclinical studies, ISM8207 demonstrated a significant synergistic anti-tumor effect, as a combination agent, against multiple solid tumor and hematologic malignancy models. It also exhibits a favorable in vitro ADME and in vivo PK profile.
Assays Completed

Enzymatic

Toxicology studies

In vitro cell-based

Developability/CMC

In vitro ADMET studies

In vivo acute efficacy studies

In vivo PK studies

In vivo PK-PD

In vivo efficacy studies with single agent

In vivo efficacy studies
Combo study with Rituximab, anti-PD-1, or paclitaxel, Azacytidine

